Development of Chrome Core® 13FM Stainless - A Soft Magnetic Stainless Steel
White Papers
Abstract
Ferritic stainless steels are being increasingly specified for automotive electromechanical devices such as fuel injectors, displacing carbon steel and silicon core irons. While the primary motivation is the need for increased corrosion resistance, ferritic stainless steels also offer higher electrical resistivity than low carbon steels and better machinability than silicon core irons.
During initial systems development, generic ferritic stainless steels such as AISI 430 were used. Demands for better and more consistent magnetic and fabrication performance have led to the development of specialized grades such as Carpenter 430FR Solenoid Quality and the Chrome Core® family of alloys.
The purpose of this paper is to identify important alloy attributes and to present the metallurgical principles that are responsible for these attributes. The development of Chrome Core 13FM stainless will be presented along with a comparison to two widely used alloys, Silicon Core Iron B-FM and Carpenter 430FR Solenoid Quality stainless.
Introduction
Material selection always involves the consideration of many material attributes. These include metallurgical characteristics as well as manufacturability and cost.
In the case of soft magnetic alloys for automotive electromechanical devices, the most important material attributes are:
Corrosion Resistance - must be sufficient to meet manufacturer's performance or warranty requirements
- Saturation induction (Bs) - ideally should be as high as possible to minimize part size and weight
- Coercive field strength (Hc) - ideally should be as low as possible to minimize "sticking" of the component. The coercive field strength is inversely proportional to the magnetic permeability (). High permeability is desirable for reducing the electrical current needed to achieve a given magnetic flux density
- Electrical Resistivity - ideally should be as high as possible to dampen eddy currents in alternating or pulsed circuits. Low eddy current losses result in a more responsive device which becomes more important as device operating speeds increase
- Ease of Fabrication - i.e., easily machined or headed
- Cost - particularly for automotive applications, alloy cost is a critical concern.
Conventional Alloys
The factors that effect material selection can best be described by first looking at the characteristics of the various types of soft magnetic alloys previously available:
Electrical Irons -- These low carbon iron alloys have been used for magnetic circuit cores, relays, and solenoids. This class of alloys is characterized by high magnetic saturation and low coercivity, which permit the design of components with low operational costs. These alloys however demonstrate the least amount of corrosion resistance, which limits the usefulness in fuel injection systems.
Silicon Irons -- The main effect of adding silicon to iron is an increase in electrical resistivity. This increased resistivity results in reduced core losses by limiting the formation of eddy currents. A secondary effect is an increase in hardness due to solid solution strengthening. These alloys possess corrosion resistance only moderately better than the electrical irons.
Chromium-Iron Magnetic Stainless Steels -- These alloys are also referred to as ferritic stainless steels and are typically composed mainly of iron and chromium. The addition of chromium to iron significantly increases the corrosion resistance and enables the use of soft magnetic alloys in corrosive environments.
The magnetic properties however are substantially reduced when iron is replaced by chromium. This results in a reduction in the magnetic saturation (the maximum magnetic flux that can be induced in the material by an applied magnetic field). The most common previously available alloy in this family is Type 430 FR which offers a useful combination of magnetic properties and corrosion resistance.
The balance of this paper will be devoted to a better understanding of the key material attributes and the metallurgical factors that influence the ferritic stainless steels. This will serve as the basis for the development of Chrome Core 13FM stainless.
Technical Attributes
An understanding of the influences of the major alloying elements on the key material attributes is needed to fully describe the relationship between alloys and properties.
Corrosion Resistance
Chromium plays the dominant role on the physical properties of ferritic stainless steel. Increasing amounts of chromium confer increasing amounts of corrosion resistance. However, the increase in corrosion resistance is not necessarily a linear function of chromium content and will vary with the corrosive medium.
An example is shown in Figure 1, which is taken from developmental work on Chrome Core alloys. Note that for this environment, a proprietary boiling corrosive water mixture, the advantage of alloys having chromium contents more than about 12% appears minimal.
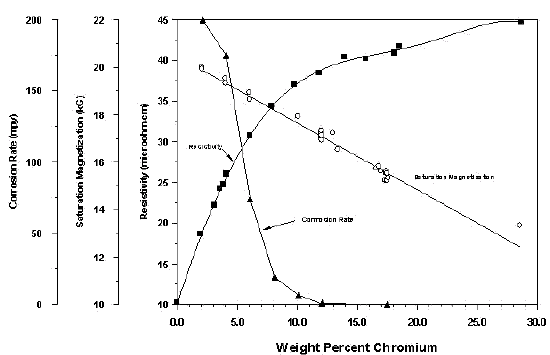
Fig 1. - Role of chromium content on corrosion resistance, room temperature electrical resistivity and saturation induction in iron-chromium alloys. The general corrosion rate was determined by immersion for 24 hours in a proprietary boiling corrosive water mixture. The electrical resistivity data are for high purity binary alloys.
Other elements that can influence corrosion resistance include molybdenum, strong carbide formers such as niobium and titanium, and sulfur. Molybdenum is added to ferritic stainless grades to improve pitting resistance. A typical level is about 0.3 % although amounts up to 2% can be used.
Strong carbide formers such as niobium and titanium are used to tie up carbon and to improve the weldability as well as the corrosion resistance. Sulfur, which is added to make free-machining grades, is detrimental to corrosion resistance creating localized attack where a sulfide inclusion meets a free surface of the part. Sulfide size and morphology may also influence corrosion resistance.
Saturation Induction
Saturation induction or saturation magnetization is the largest magnetic flux density induced in a material and represents the ideal state wherein all the elementary magnetic moments are aligned by the applied magnetic field. Since the force of attraction varies as the square of the flux density induced in the part, there is a great advantage to using high saturation alloys.
In ferritic stainless steels, the saturation induction is a function of the nonmagnetic element content. Since the atomic moment of iron, the main ferromagnetic constituent, is being effectively diluted by the nonmagnetic constituents, the saturation induction should exhibit a linear decrease with increasing chromium content.
This is also shown in Figure 1. Note that other elements such as manganese, silicon and molybdenum will also reduce the saturation magnetization in proportion to their content. Because low saturation inductions are undesirable, there is clearly a tradeoff between corrosion resistance and saturation induction that should be recognized when choosing an alloy for a particular application.
Coercive Field Strength
Unlike nickel-iron and iron-cobalt soft magnetic alloys, there does not seem to be a "special" composition in the iron-chromium system that results in exceptionally high permeability or exceptionally low coercivity. Such special compositions, such as 80% nickel-iron or 50% iron-cobalt, are typically associated with a minimum in magnetocrystalline anisotropy.
In ferritic stainless steels, the technological magnetic properties such as permeability and coercive field strength are strongly related to the microstructure of the alloy rather than the chemistry. These factors include grain size, inclusion content, carbide and nitride precipitates and grain orientation. The effect of grain size on coercivity is shown in Figure 2.
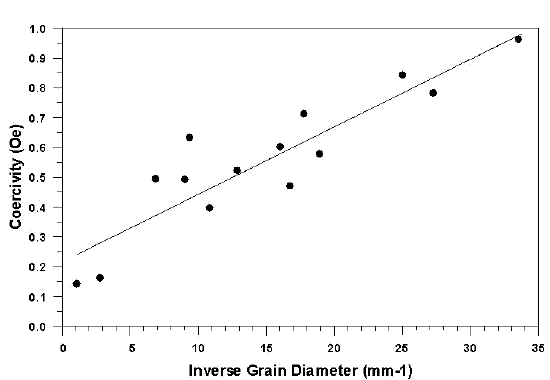
Fig 2. - Role of grain size on coercivity in an experimental 12% Cr-Fe alloy.
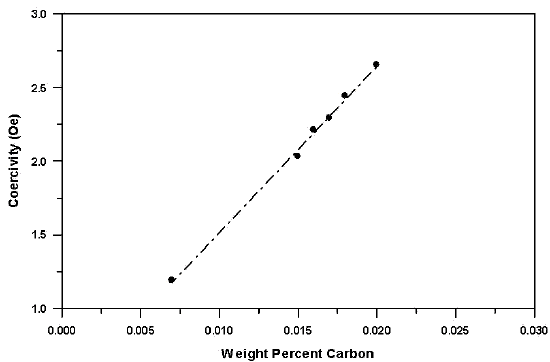
Fig 3. - Role of carbon content on coercivity in experimental 12% Cr-Fe alloys. width="545" xpos="0"> In free-machining grades, the sulfur content may be the ultimate controlling factor in the attainment of good magnetic performance.
First, the sulfide inclusions can inhibit grain growth. As noted earlier, smaller grain size will result in increased coercivity. Second, the difference in thermal expansivity of the sulfides and the matrix can result in strains upon cooling from the heat treatment temperature. Finally, since the sulfide is effectively a nonmagnetic cavity, reverse magnetic domains will be formed increasing the difficulty of magnetizing the alloy.
A general relationship between microstructure and coercive field strength is:
![]()
where Hco denotes an intrinsic value related to the magnetocrystalline anisotropy, p denotes the dislocation density and is a measure of the amount of cold work or strain, d denotes the grain size and Nv denotes the density of inclusions whose dimensions are comparable to the magnetic domain wall width.
The above equation indicates that for lowest coercive field strength it is necessary to have no residual cold work, large grain size and freedom from fine particles that can pin magnetic domains. This is an obvious impossibility when sulfides or carbides are present. Carbon appears particularly detrimental to the achievement of good soft magnetic properties as shown in Figure 3.
Electrical Resistivity
The electrical resistivity in ferritic stainless steels appears to be dictated by alloy composition. The role of chromium on the resistivity is shown in Figure 1. The data on the high purity binary alloys are from Adcock.[1] Note that most of the increase in electrical resistivity occurs between 0 and 12% chromium for both the commercial alloys and the high purity binary alloy.
To further increase the electrical resistivity it is necessary to add other elements. Silicon and aluminum are the most effective additions. This is shown in Figure 4 for nominally 12% chromium-iron alloys. The highest resistivities shown are for Carpenter 1JR® type alloys.
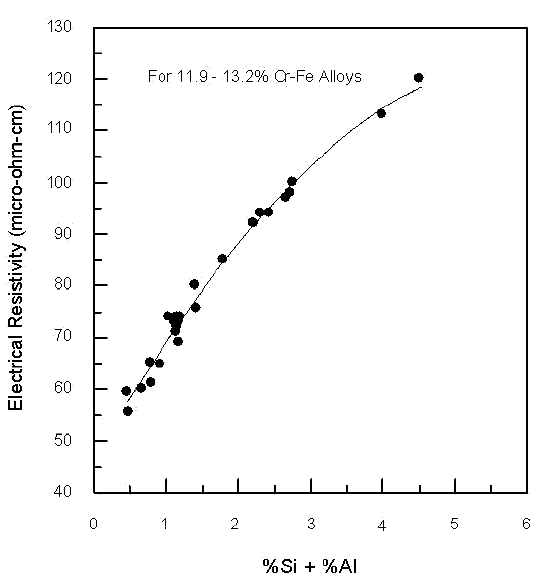
Fig 4. - Influence of silicon and aluminum on the room temperature electrical resistivity of 12% Cr-Fe alloys. Fabrication
A key consideration for these alloys is the ease of fabrication, which has historically translated to improved machinability. Although some components are capable of being produced by cold or warm heading, it has been our experience that some amount of machining is involved in almost all parts production.
In addition, some parts require welding. Machinability, headability and weldability impose different constraints on alloy chemistry and processing.
Machinability is a complex subject[2]. Apart from the obvious desire for high metal removal rates, other issues such as surface finish, tool wear and suitability for other operations such as welding are important and influence the alloy selection process. Metal removal during machining occurs by repeated localized fracture.
Improved machinability is obtained when the alloy chemistry and structure promote this process. This is accomplished by lowering the work hardening rate of the metal matrix and/or by creating heterophase interfaces by use of free-machining additives that are sites for microcrack initiation. The latter is more effective and has been the focus of most alloy development work.
Free-machining additives consist of sulfur, selenium, tellurium, lead, bismuth, phosphorus and certain "soft oxides." Other factors influencing the machinability include grain size, hardness and interstitial elements.
Sulfur is the most common free-machining additive and exists as manganese-chromium-iron sulfide inclusions. Amounts up to 0.5% have been used, although amounts of 0.25-0.35% are more common. Apart from the amount of sulfur, the manganese to sulfur ratio and the sulfide size and shape are important.
Improved machinability is favored by a high manganese to sulfur ratio and large, globular sulfides. High sulfur contents result in impaired magnetic performance, corrosion resistance, headability and weldability so despite its widespread use; sulfur is often not the best free-machining additive.
Selenium forms a manganese selenide (MnSe) inclusion but is less effective than sulfur on an equivalent weight basis. However, selenium reportedly provides a better surface finish and may result in improved cold selenium is to promote globularization and enlargement of sulfide inclusions. Carpenter currently produces some Se containing grades, although none are routinely used in magnetic applications.
Tellurium forms a manganese telluride (MnTe) inclusion and is reportedly more effective than sulfur in free-machining austenitic stainless steels. Like selenium, it also promotes globularization and enlargement of sulfide inclusions.
Carpenter has conducted research on Te additions to austenitic and martensitic stainless steels and has patents issued on such additions. However, Te causes hot working problems in austenitic stainless steels and has not been used in commercial practice by us.
Lead and bismuth are reported to be the best free-machining additives leading to high metal removal rates, superior surface finishes and lower tool wear. Moreover, lead has been shown to result in better corrosion resistance and cold headability than sulfur.
Both elements are essentially insoluble in stainless steels that complicates their addition to the molten alloy. These elements are added in either the ingot mold or concasting tundish and achieving a uniform distribution of lead or bismuth particles in the steel is an ongoing problem. Lead is also toxic a factor that limits its attractiveness and both elements can promote hot working difficulties.
Phosphorus is used as a free-machining additive in carbon and alloy steels, such as Silicon Core Iron B-FM. Unlike the other elements mentioned, phosphorus does not form inclusions; rather it lowers the strain-hardening rate of the matrix aiding in chip formation. Although it has been used in stainless steels, it has some undesirable effects on corrosion resistance and is not commonly used today.
Certain calcium - aluminum - silicon oxides (silicates), formed during calcium deoxidation of steels, soften at the high temperatures encountered at the tool-metal interface. The soft oxide can coat the tool surface thus lubricating it and reducing tool wear.
This technique was discovered by and is used in the carbon and alloy steel industry. It has also been used in stainless steels. One limitation is the need to maintain a balance between the metallic constituents of the oxide otherwise hard, abrasive oxides can be formed decreasing tool life.
Development of Chrome Core 13FM Stainless
For many automotive applications, the corrosion environment is such that 16-18% Cr is not required hence the development of lower chromium content alloys such as Chrome Core 8-FM and 12-FM alloys. Lowering the chromium content reduces the alloy cost slightly but, what is more important, increases the saturation induction.
The development of these alloys was motivated by the desire to directly replace silicon iron components. Since the parts were originally designed to take advantage of the high saturation induction of the silicon iron alloys, the use of 16-18% Cr alloys, with their lower saturation induction, would have required a redesign.
Moreover, the environment, alcohol based fuel, was sufficiently benign that increasing the Cr content from 12 to 18% would have little benefit to corrosion resistance. Both Chrome Core 8-FM and 12-FM alloys have been commercially available for a number of years and have been used in automotive electromechanical components.
Despite the success of these alloys, there has been a continuing need for improved device performance without sacrificing machinability and corrosion resistance. Chrome Core 13FM stainless was developed with these in mind.
The principal improvements obtained in Chrome Core 13FM stainless are higher electrical resistivity and lower coercivity. The increased electrical resistivity was accomplished by a slight increase in chromium content from Chrome Core 12FM alloy and by increasing the silicon content to about 1.5%. Apart from increasing the electrical resistivity, the higher silicon content suppresses the formation of austenite, allowing for higher heat treating temperatures.
The improved soft magnetic properties were achieved by further reductions in the carbon and nitrogen contents and by modification of the bar processing. To maintain adequate machinability, relatively high sulfur contents are used. As mentioned previously, the sulfides impede grain growth leading to grain sizes of typically about ASTM 7 to 8 in heat treated parts.
Increasing the annealing temperature or time results in virtually no increase in grain size or improvement in magnetic properties. It was found, however, that by cold drawing previously annealed bar by a small amount, that strain induced grain boundary migration would occur during the final heat treatment. Because the driving force is strain rather than the reduction in surface area associated with normal grain growth processes, the pinning effect of the sulfides was reduced.
This leads to larger grain sizes and, therefore, higher permeability and lower coercivity. The influence of the degree of cold work on coercivity after the final heat treatment is illustrated in Figure 5. Note that a minimum in coercivity occurs around 15% for this particular experimental alloy. The actual strains needed for lowest coercivities after heat treatment will depend on prior processing and bar diameter.
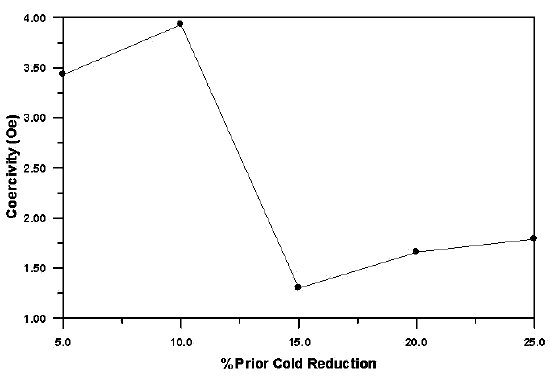
Fig 5. - Influence of cold reduction prior to the final anneal on the coercivity of an experimental 13% Cr-Fe alloy. Note that a minimum value of coercivity exists for a critical reduction of about 15%.
Comparison of Alloys
The properties of Chrome Core 13FM stainless are shown in Table 1, along with typical properties for heat treated Silicon Iron B-FM, Carpenter 430FR Solenoid Quality Stainless, and other Chrome Core alloys.
Note that while Silicon Iron B-FM has superior soft magnetic performance, i.e. higher saturation magnetization and permeability, it exhibits virtually no useful corrosion resistance, has lower electrical resistivity and inferior machinability.
The principal distinction between Chrome Core 13FM stainless and Carpenter 430 FR Solenoid quality is the saturation magnetization. It should be noted that the data for 430FR and the other Chrome Core alloys reflect the use of the same controlled bar processing developed for Chrome Core 13FM stainless, so that the differences in the permeability and coercivity are very minor. 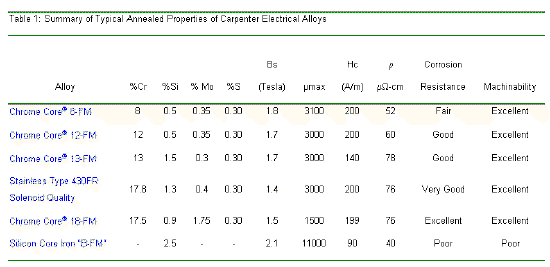
Conclusion
Many factors play a role in selecting soft magnetic alloys. The goal is to select an alloy that provides the best combination of properties for the application. In the case of automotive fuel injectors, the maximum attainable magnetic saturation and the lowest coercivity in desired in combination with adequate corrosion resistance. Carpenter Chrome Core 13FM stainless can be considered a candidate to provide the next step in the optimization of this combination of properties.
References
- F. Adcock, "Chromium-Iron Constitutional Diagram", JISI, vol. 124, pp. 99-149, 1931
- Kosa, T. and Ney, R. P., Sr., "Machining of Stainless Steels", Metals Handbook - Ninth Edition - vol. 16 - Machining, Metals Park, OH ASMI 1989, pp. 681-707.
***
By Daniel A. DeAntonio and M. Scott Masteller
Carpenter Technology Corporation
Reading, PA
USA