Ideas for Improving Machinability, Productivity and Profitability of Stainless Steel
White Papers
Passivating and Electropolishing Stainless Steel Parts
Parts and components machined from stainless steels must be passivated to maximize their essential corrosion resistance. Good passivating practice can make the difference between satisfactory performance and premature failure. Poor practice can actually cause corrosion.
 |
| Two small instruments for orthopedic surgery made from Carpenter Custom 455® stainless and CarTech Custom 630 (17Cr-4Ni) stainless. Each bright electropolished instrument contrasts with identical instruments covered by brown oxide from prior heat treatment. Control of finish between bright and dull can be maintained by judicious use of abrasive wheel and fine glass bead blasting. (Photo courtesy of Troy Innovative Instrument, Inc., Middlefield, OH) |
The process of passivation is sometimes misunderstood. It is not a scale removal treatment, nor anything similar to a coat of paint. It is a post-fabrication method of maximizing the inherent corrosion resistance of the stainless steel from which the workpiece was made.
Not everyone agrees on the precise mechanics of how passivation works. It is certain, however, that a protective oxide film exists on the surface of passive stainless steel before it is fabricated. This invisible film is extremely thin, about 100,000 times thinner than a human hair!
Clean, freshly machined, polished or pickled stainless steel parts automatically acquire this protective film from exposure to oxygen in the atmosphere. Under the best conditions, this oxide film covers all part surfaces.
The need for passivation arises when parts are fabricated, either by cold forming or machining. Contaminants such as shop dirt or iron particles from cutting tools may be transferred to the surface of the stainless steel parts during fabrication. These foreign particles can reduce effectiveness of the original protective film. If they are not removed, corrosive attack may begin.
Under certain conditions, rust spots may appear on machined parts. This is actually corrosion of foreign particles from the tool steel, not the parent metal. Sometimes the crevice at the embedded tool steel particle or its corrosion products may cause attack of the part itself.
Likewise, small particles of iron-containing shop dirt may stick to the stainless part surface. Although the metal may appear shiny in the as-machined condition, the invisible particles of free iron can cause rusting on the surface after exposure to the atmosphere.
Exposed sulfides, if ignored, also can be a problem. They derive from the addition of sulfur to stainless steels to improve machinability. Sulfides improve the alloy’s ability to form chips that break away cleanly from the cutting tool during the machining process. If the part is not properly passivated, sulfides can act as initiation sites for corrosion on the surface of the fabricated product.
A two-step procedure can provide the best possible corrosion resistance. First is cleaning, a fundamental, but sometimes overlooked procedure. Second is passivating treatment in an acid or electrochemical bath.
Cleaning
Grease, coolant or other shop debris must be thoroughly cleaned from the surface to obtain the best possible corrosion resistance. Machining chips or other shop dirt can be wiped carefully off the part. A commercial degreaser or cleanser may be used to clean off machining oils or coolants. Foreign matter such as thermal oxides may have to be removed by grinding, or by methods such as acid pickling.
Occasionally, a machine operator might skip the cleaning, falsely assuming that by immersing a grease-laden part in an acid bath, both cleaning and passivating will take place at the same time. That doesn’t happen. Instead, the contaminating grease reacts with the acid to form gas bubbles. These bubbles collect on the surface of the workpiece and interfere with passivation.
 |
| High strength, pin-like surgical instruments made from CarTech Custom 455® stainless just removed from electropolishing tank. After a deionized water rinse, the instruments will have a lustrous finish. (Photo courtesy of Troy Innovative Instrument, Inc., Middlefield, OH) |
Even worse, contamination of the passivating solution, sometimes with high levels of chlorides, can cause “flash attack.” Instead of obtaining the desired oxide film with a shiny, clean, corrosion-resisting surface, the flash attack produces a heavily etched or darkened surface. This is a deterioration of the very surface that passivation is designed to optimize.
Martensitic/Precipitation Hardened Stainless Grades
Parts made from martensitic stainless steels (CarTech 410, CarTech 420 and CarTech 440 series) are magnetic, with moderate corrosion resistance and high yield strengths. These alloys are hardened at a high temperature, then tempered to obtain the hardness and mechanical properties desired.
Precipitation hardenable (PH) stainless steels ( CarTech Custom 630 (17Cr-4Ni) ,CarTech Custom 450 , CarTech Custom 455 and CarTech Custom 465 stainless) offer a better combination of strength and corrosion resistance than the martensitic alloys. These PH grades are rough machined, aged at lower temperatures, then finish machined.
Workpieces made from the preceding stainless steels must be thoroughly cleaned with a degreaser or cleanser to remove any traces of cutting fluid before heat treating. Otherwise, cutting fluid remaining on the parts will cause excessive oxidation. This condition can result in undersize parts with a pitted finish after the scale is removed by acid or abrasive methods. If cutting fluids are left on parts that are bright hardened, as in a vacuum furnace or protective atmosphere, surface carburization may occur, leading to a loss of corrosion resistance.
Passivating - Nitric Baths
After the stainless steel parts or components are thoroughly cleaned, they are ready for immersion in a passivating bath. More corrosion-resistant, chromium-nickel stainless steels can be passivated in a 20% by vol. nitric acid bath (Fig. 1).
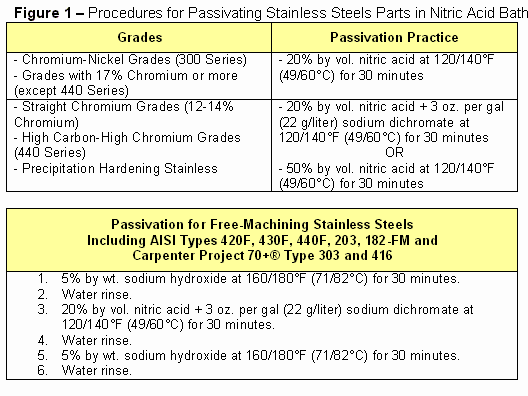
As shown in the same table, less resistant stainless grades – straight chromium, high carbon-high chromium and precipitation hardened martensitic grades – can be passivated by adding sodium dichromate to the nitric acid bath to make the solution more oxidizing and capable of forming a passive film on the surface.
Another option is to increase the concentration of nitric acid to 50% by vol. The sodium dichromate addition and the higher nitric acid concentration both reduce the chance of undesirable flash attack. It should be understood here that the 50% nitric acid concentration is more oxidizing and therefore less aggressive to stainless steels than 20% nitric acid.
Free-Machining Stainless Steels
The procedure for passivating free-machining stainless steels (Fig. 1) is somewhat different from that used with non-free-machining stainless alloys. That is because the sulfides of sulfur-containing free-machining grades are partially or totally removed during passivation in a typical nitric acid bath, creating microscopic discontinuities in the surface of the machined part. Even normally efficient water rinses can leave residual acid trapped in these discontinuities after passivation. Unless this acid is neutralized or removed, it can then attack the surface of the part.
To effectively passivate the free-machining stainless steels, Carpenter has developed the A-A-A (alkaline-acid-alkaline) process which neutralizes trapped acid. This passivation method can be done in less than two hours, as follows:
(a) After degreasing, soak the parts for 30 minutes in a 5% solution of sodium hydroxide at 160ºF to 180ºF (71ºC to 82º). (b) Rinse the parts thoroughly in water (c) Immerse the parts for 30 minutes in a 20% by vol. nitric acid solution containing 3 ounces per gallon (22g/liter) of sodium dichromate at 120ºF to 140º (49º to 60ºC). (d) Remove the parts from bath and flush with water. (e) Immerse the parts in the sodium hydroxide solution for another 30 minutes. (f) Water rinse the parts again and dry them, completing the process.
The benefits of using the alkaline-acid.alkaline method are demonstrated in Fig. 2. The left test cone clearly shows the improved surface and resistance of free-machining stainless steel when passivated by the A-A-A method. The visible result of conventional passivation can be seen in the right test cone.
Fig. 2 – Left test cone is passivated using the A-A-A method. Results of conventional passivation shown on right. Both were exposed to salt spray.

Passivating - Citric Baths
Citric acid passivation has been gaining favor with fabricators who wish to avoid the use of mineral acids or solutions containing sodium dichromate, and elude the disposal problems and greater safety concerns associated with their use. Citric acid is considered environmentally friendly in every respect. It is on the GRAS (Generally Regarded As Safe) list compiled by the FDA as a material that is safe for people to handle.
While citric acid passivation offers attractive environmental advantages, shops might want to continue the course if they are having success with mineral acid passivation and are having no safety issues. There may be no compelling need for those fabricators to change if they have a clean shop, well maintained and clean equipment, coolant free of iron-containing shop dirt and a process that yields good results.
Passivation in citric acid baths has been found useful for a large number of stainless steel families, including several individual stainless grades (Fig. 3). The conventional nitric acid passivation methods from Fig. 1 are included for convenience. Observe that the older nitric acid formulations are in volume percent, while newer citric acid concentrations are in weight percent. It is important to note that, in implementing these procedures, a careful balance of immersion time, bath temperature and concentration is critical to avoid the “flash attack” described earlier.
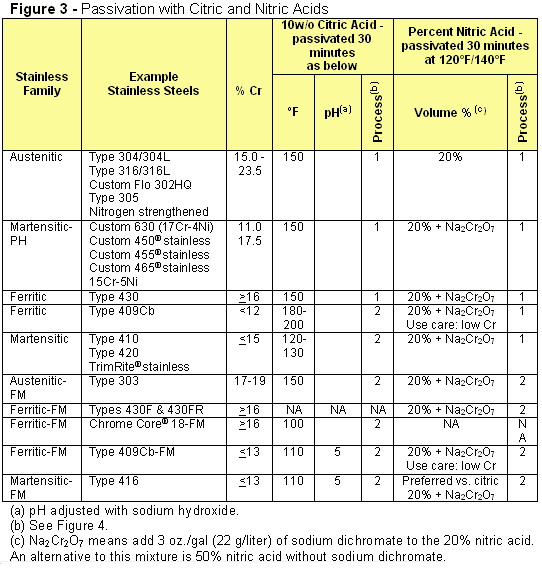
The passivation treatment varies depending on chrome content and machinability characteristics of the grades in each family. Note the columns referring to Process 1 or Process 2 in Fig. 4. Process 1 includes five steps: clean and degrease, rinse, passivate, rinse and dry. Process 2 includes seven steps: clean and degrease, rinse, passivate, rinse, neutralize, rinse and dry.

Laboratory tests have indicated that citric acid passivating procedures were more prone to “flash attack” than nitric acid procedures. Factors causing this attack included excessive bath temperature, excessive immersion time and bath contamination. Citric acid products containing corrosion inhibitors and other additives (e.g. wetting agents) are commercially available that reportedly reduce sensitivity to “flash attack”.
The ultimate choice of passivation will depend on the acceptance criteria imposed by the manufacturer for whom the parts or components are to be made. For more information, refer to ASTM A 967 “Standard Specification for Chemical Passivation Treatments for Stainless Steel Parts.” The specification can be accessed at www.astm.org
Electropolishing
Electropolishing is an electrochemical process used to polish a metal surface by removing a microscopic amount of material from the workpiece. It is a frequently used method that more accurately conditions, rather than passivates the surface of a part. A part that has been electropolished requires no subsequent passivation.
This is the process of choice for parts and components that must have a very smooth, lustrous, mirror finish such as that required for surgical instruments. For bright-finish parts that reflect too much glare – as in the surgical suite – glass beads or a fine abrasive buffing pad may be used to tone down the surface finish.
Electropolishing is accomplished by creating an electrochemical cell in which the material to be polished is the anode (stainless steel part). A cathode is formed to duplicate the geometry of the part surface. The anode and cathode are submerged in a heated electrolyte bath. For stainless steels, a variety of mixed acid solutions is suggested for the electrolyte bath, including glycolic, phosphoric and sulfuric. When a DC current is applied, an electrical charge dissolves metal ions from the part surface.
Since electropolishing removes surface material and contaminants, it improves the corrosion resistance of stainless parts so processed. In addition, electropolishing deburrs as it polishes the surface. Finally, the improved microfinish reduces product adhesion and contamination buildup.
Testing Treated Parts
Tests can be performed to determine how effective passivation has been in removing free iron and other exogenous matter from treated surfaces. These tests can be used on electropolished surfaces as well. Each of five test methods are described in detail in the previously mentioned ASTM A 967 specification. They are known as: A – water immersion test, B – high humidity test, C – salt spray test, D – copper sulfate test and E – potassium ferricyanide-nitric acid test.
It is essential that the test method employed is matched to the grade to be evaluated. A test that is too severe may reject perfectly good material, while one that is too lenient may accept unsatisfactory parts.
The 400 series precipitation-hardening and free-machining stainless steels are best evaluated in a cabinet capable of maintaining 100% humidity (samples wet) at 95ºF (35º) for 24 hours. The cross section is usually the most critical surface, particularly for free-machining grades. One reason for this is that the sulfides, elongated in the direction of working, intersect this surface.
Critical surfaces should be positioned upward, but at 15 to 20 degrees from the vertical to allow any moisture to run off. Material that has been properly passivated will be virtually free of rust, although it may show some light staining.
Austenitic, non-free-machining stainless steels also may be evaluated by means of a humidity test. When so tested, liquid droplets of water should be present on the surface of samples, revealing free iron by the presence of rust formation.
A faster method is available using a solution from ASTM A380, “Standard Recommended Practice for Cleaning and Descaling Stainless Steel Parts, Equipment and Systems.” This test consists of swabbing the part with a copper sulfate/sulfuric acid solution, maintaining wetness for six minutes and observing whether there is any plating of copper.
Alternatively, the part may be immersed in the solution for six minutes. Copper plating occurs if iron is dissolved. This test should not be applied to surfaces of parts for use in food processing. Also, it should not be used for the martensitic or lower-chromium ferritic stainless steels of the 400 series because false-positive results are likely.
Historically, the 5% salt spray test at 95ºF (35ºC) also has been used to evaluate passivated samples. This test, too severe for some alloys, generally is not necessary to confirm that passivation has been effective.
 |
| Stainless steel components for the handle of a surgical instrument after they have been heat treated and passivated in this citric acid-base solution. (Photo courtesy of Troy Innovative Instrument, Inc., Middlefield, OH) |
To help you with the passivation process, here is a checklist of good procedures and a second list of potential mistakes to be avoided.
Best Practice
- Clean first, removing all particles of oxide or heat tint before passivating.
- Assign certain machines to fabricate stainless steels only. Stay with the same preferred coolant to cut stainless steels, to the exclusion of all other metals.
- Rack parts individually for treatment to avoid metal-to-metal contact. This is especially important with free-machining stainless steels where free flow of passivating and rinse solutions is needed to diffuse away corrosion products from sulfides and avoid pockets of acid.
- Avoid chlorides which, in excess, can cause harmful flash attack. When possible, use only a good grade of water containing less than about 50 parts per million (ppm) of chlorides. Tap water is usually adequate, and in some cases up to several hundred ppm chlorides can be tolerated.
- Replace baths on a regular schedule to avoid a loss in passivation potential that can result in flash attack and ruined parts. Baths should be maintained at proper temperature because out-of-control temperature may allow localized attack.
- Maintain very specific schedules for solution replacement during high production runs to minimize the possibility of contamination. Use a control sample to test the bath’s effectiveness. If the sample is attacked, it is time to change the bath.
Mistakes to Avoid
- Do not passivate stainless steel parts that have been carburized or nitrided. Parts so treated may have their corrosion resistance reduced to the point where they are subject to attack in the passivating tank.
- Don’t use tooling with iron content (floor, equipment, coolant, etc.) that is not exceptionally clean. Steel grit can be avoided by using carbide or ceramic tools.
- Don’t forget that attack can occur in a passivating bath if parts are improperly heat treated. High-carbon, high-chromium martensitic grades must be hardened to become corrosion-resistant. Passivation is frequently performed after a subsequent temper using a temperature that maintains corrosion resistance.
- Don’t overlook the nitric acid concentration in the passivating bath. It should be checked periodically using a simple titration procedure below.
- Don’t passivate more than one stainless steel at a time. This discipline can prevent costly mixups and avoid galvanic corrosion.

***
By John H. Magee, Specialist – Stainless Alloy R&D, and Robert K. Mohr, Manager, Regional Metallurgy
Carpenter Technology Corporation
Wyomissing, PA, USA