Selecting Alloys for Severely Corrosive Environments
Technical Guide
Parts and components exposed to aggressive environments in the chemical process industries require corrosion resistance greater than that provided by conventional stainless steels.
Indeed, nowhere else is a metal's reaction to its environment more intense than in the harsh chemicals, elevated temperatures and high pressures encountered in chemical processing. Here, pipes, pumps, valves, vessels and other metallic structures must survive long term exposure to conditions ranging from mildly to severely corrosive, to downright insidious.
Since corrosive chemicals cannot be eliminated from process applications, their attack must be controlled by materials of construction such as stainless steels and other highly-alloyed metals.
No single alloy, of course, can be suitable for every application. Once the alloys with the desired corrosion resistance have been identified, then requirements for strength, fabrication and availability must be addressed.
The guidelines and data to follow are intended to help you determine, in general, which alloys might be considered for various corrosive environments. None of this information is meant to replace the technical expertise of a corrosion engineer. However, it can serve as a starting point.
Alloying Elements
Increased levels of elements such as nickel, molybdenum, copper and chromium may be used to provide resistance to the severely corrosive conditions found in some chemical process and oil field environments.
Many highly alloyed materials can be viewed as extensions of the austenitic stainless steels. Several are hardenable only by cold working. Others have columbium, titanium, aluminum and/or copper additions to make them age hardenable by heat treatment.
Typically, stainless steels are alloys of iron to which a minimum of 11% chromium has been added to provide a passive film that resists "rusting" when the material is exposed to the weather. To obtain greater corrosion resistance, more chromium is added to produce stainless grades with chromium contents of 15%, 17%, 20% and higher. Chromium provides resistance to oxidizing environments, such as nitric acid, and to pitting and crevice attack.
Other alloying elements can be added for improved corrosion resistance or higher strength. Molybdenum is probably the most effective element for improving resistance to pitting and crevice corrosion.
Nickel provides resistance to reducing environments and, above 25%, improves resistance to stress corrosion cracking. Nickel and nitrogen typically retard the formation of sigma phase, an intermetallic which can have a harmful effect on corrosion resistance and mechanical properties.
Nitrogen may be added to increase strength and improve corrosion resistance, especially to pitting or crevice attack. Higher levels of elements such as chromium and manganese increase nitrogen solubility during casting.
Copper addition improves resistance to general corrosion in sulfuric acid, and strengthens some precipitation-hardenable grades. Pitting resistance, however, can be reduced in some alloys as the copper content increases.
Columbium and titanium tend to stabilize carbon, thus minimizing formation of chromium carbides which can impair corrosion resistance. Along with aluminum, these elements also can be used to age harden iron- and nickel-base alloys.
Environment and Application
In addition to alloy composition, other factors like service environment and product application have a bearing on corrosion resistance. The following conditions, for example, should be considered carefully before making an alloy choice:
- The bulk solution composition, including the solution pH and concentration of its components.
- Temperature of the corrodent. In general, the corrosion rate increases with increasing temperature. Some metals that provide good service at low temperatures are unsuitable for the same solutions at more elevated temperatures.
- Source of the heat. If heat is transferred from the metal to the solution, the corrosion rate may be increased due to the higher metal temperature.
- Impurities in the corrosive medium. The presence of even minor amounts of impurities can alter the corrosion rate significantly. While chlorides are expected to increase attack, some impurities actually can reduce the corrosion rate.
- Adherent deposits. Deposits on the metal surface can cause crevice corrosion, particularly in the presence of chloride ion impurities.
- Degree of aeration. Liquids kept in closed containers, without air or air circulation, may cause different degrees of corrosion than that occurring when the same liquids are freely exposed to the air. Similarly, the presence of atmosphere gases can change the corrosive behavior of metals to a marked degree.
- Velocity of the corrodent may affect both the type and severity of corrosion.
It should be noted that published corrosion tables usually address only the bulk solution composition and fluid temperatures. Thus, the information found there should be tempered with the other considerations just mentioned.
Other Considerations
Mechanical properties, usually strength, often play a critical role in the selection of a corrosion resistant alloy (Fig. 1). Depending on the application, the specifier may be concerned primarily with the material's hardness, impact resistance, fatigue strength or stress rupture resistance - or perhaps more than one of these properties.
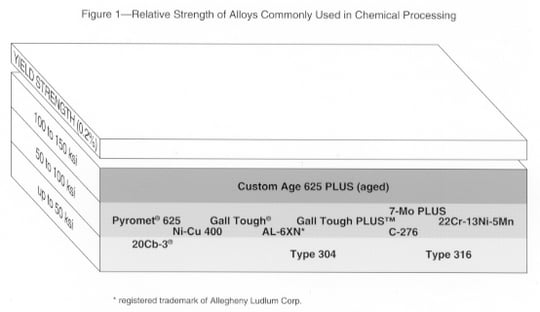
When selecting an alloy based on the desired mechanical property, consideration should be given the potential effect of that property on the material's corrosion resistance. Typically, alloys that have been heat treated or cold worked to obtain a higher strength level are more susceptible to stress corrosion cracking.
Material condition can be important. A smooth surface finish often improves corrosion resistance. This is especially true for lower alloy stainless steels in severe atmospheric exposures. For optimum corrosion resistance, surfaces must be free of scale and foreign particles. Also, finished parts should be passivated in accordance with alloy producer recommendations.
Metallurgical condition of the alloy should be considered. Corrosion resistance, particularly to stress-corrosion cracking, can depend on whether an alloy is annealed, hardened or cold worked.
Both design and method of fabricating the alloy are important. The key principle in designing a part is to avoid crevices and contact between two dissimilar metals.
Fabrication should be considered from two perspectives - (a) how easy or difficult it is to machine, cold form, weld and heat treat the material, and (b) what effect the fabrication processes will have on the material's corrosion resistance. Sometimes it is necessary to modify the intended fabrication procedures to correct or prevent potential corrosion problems.
Thermal treatment in fabrication can be a determining factor. Welding or stress relieving, for example, can affect resistance to corrosion, particularly to stress corrosion cracking and intergranular corrosion.
Alloys for Severe Corrosives
A good selection of alloys is available for service in highly corrosive environments. Their typical chemical compositions are shown in Fig. 2. As a group, the high-alloy austenitic stainless steels provide resistance to general corrosion and several forms of localized attack, along with high levels of ductility and toughness.
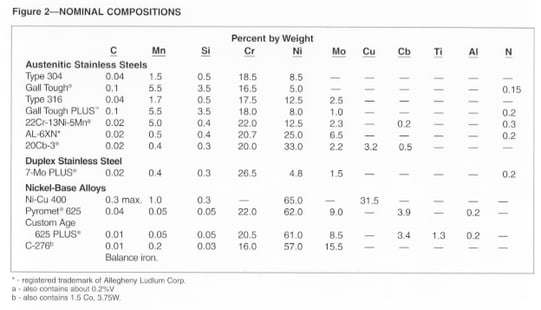
Duplex 7-Mo PLUS® stainless (UNS S39295) provides excellent resistance to oxidizing media and localized corrosion, and has about twice the yield strength of the typical austenitic stainless steels.
The nickel-base alloys provide resistance to very severe environments. For example, Carpenter Technology Custom Age 625 PLUS® alloy (UNS N07716) can be considered for service in elevated-temperature oil field environments which may contain brine, hydrogen sulfide, carbon dioxide and elemental sulfur.
A method has been developed to help in the selection of alloys for severely corrosive service. The charts in Figs. 3 through 8 show alloys' susceptibility to attack in pertinent environments or by specific forms of corrosion.
The typical strength level is plotted on the horizontal axis, but other strengths can be obtained for each alloy by varying the processing. Annealed strength levels are used in most cases, except for Carpenter Technology Custom Age 625 PLUS® alloy, where the more typical aged condition is used.
Resistance to each form of corrosion is also plotted based on material in the annealed or annealed-plus-aged condition. When material is used in other conditions (for example, as-welded) care must be taken to assure that corrosion resistance and mechanical properties are maintained.
Where to Start in the Selection Process
Carpenter Technology 304 stainless steel can be considered the baseline alloy because it is the most widely used 18-8 stainless alloy. It is an austenitic stainless that is resistant to sterilizer solutions, most organic chemicals and dyestuffs, and a wide variety of inorganic chemicals. It should be considered only for the milder media found in the chemical process industry.
Carpenter Technology Gall-Tough® stainless (UNS S20161) has corrosion resistance similar to that of Carpenter Technology 304, along with superior self-mated galling resistance and metal-to-metal wear resistance. It can be considered for parts that are in relative motion under load, without lubricants.
For greater resistance to corrosion, Carpenter Technology 316 stainless steel may be the answer. It resists the attack of phosphoric acid, sulfite liquors and sulfurous acids. This austenitic stainless has been used for equipment in the process industries to produce inks, rayon, photographic chemicals, paper, textiles, bleaches and rubber.
Carpenter Technology Gall-Tough PLUS® stainless, offering higher strength and corrosion resistance comparable to that of Carpenter Technology 316 stainless, is a new alloy that also provides outstanding self-mated galling and metal-to-metal wear resistance.
Sulfuric Acid
Sulfuric acid is a common environment that typically causes general corrosion. Fig. 3 provides an overall ranking of several materials in pure sulfuric acid.
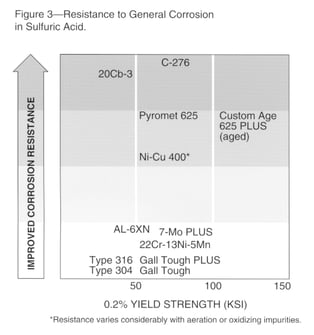
Severity of attack in sulfuric acid varies widely with the temperature, concentration and aeration of the acid. Aeration is generally beneficial to higher chromium materials and deleterious to nickel-copper alloys. Increased velocity usually increases attack of all alloys.
Impurities such as iron, copper and chromium ions reduce attack of stainless steels in sulfuric acid. Although oxidizing impurities are beneficial for stainless steels, they are deleterious for nickel-copper alloys.
Impurities such as chlorides often result in increased general attack, and can cause pitting or stress-corrosion cracking. Materials with insufficient nickel content can also experience cracking in pure sulfuric acid. It is usually wise to confirm that an alloy chosen for resistance to general corrosion does not exhibit stress-corrosion cracking in the environment of interest.
Generally, Cr-Ni-Mo-Cu alloys like Carpenter Technology 20Cb-3® stainless (UNS N08020), with nickel content above about 32%, can be considered for resisting cracking and general corrosion in a wide variety of sulfuric acid environments.
Several materials in Fig. 3 can be considered for specific sulfuric acid environments. Conventional stainless grades such as Carpenter Technology 316 should be considered for only very dilute or highly concentrated acid at low temperatures. Slightly improved resistance in these concentration ranges may be obtained with higher chromium materials such as 7-Mo PLUS® stainless or Carpenter Technology 22Cr-13Ni-5Mn (UNS S20910) stainless steels.
Better resistance to intermediate concentrations may be obtained with alloys having higher nickel, such as AL-6XN stainless (UNS N08367) and, particularly, Carpenter Technology 20Cb-3® stainless steel. Carpenter Technology Nickel-Copper 400 alloy (UNS N04400) can be considered for air-free sulfuric acid environments, but it is readily attacked in air-saturated solutions.
Nitric Acid
Nitric acid is another example of an environment that can cause general corrosion. Since this acid is oxidizing, alloys with higher chromium are expected to provide improved resistance. Materials that may be considered for nitric acid service are ranked in Fig.4.
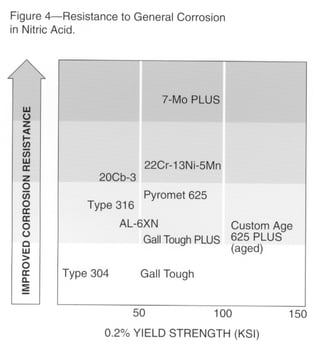
Carpenter Technology 316 stainless has useful resistance, Carpenter Technology 22Cr-13Ni-5Mn is highly resistant, and 7-Mo PLUS stainless may be considered for the most severe applications. Materials with lower chromium have very limited usefulness, and alloys such as Carpenter Technology Nickel-Copper 400 alloy are rapidly attacked.
Nitric acid also can cause intergranular attack of sensitized materials, due to precipitation of carbides or sigma phase at the grain boundaries. Because of this tendency, the Huey test (ASTM A262, boiling 65% nitric acid) is usually employed to screen materials prior to nitric acid service.
Pitting and Crevice Corrosion
Alloys that resist pitting and crevice corrosion are shown, with their ranking, in Fig. 5. This figure may apply in many environments where chloride pitting rather than general corrosion is expected to occur. One example is seawater.

Increased chromium and molybdenum improves resistance of stainless steels to pitting and crevice attack. Nitrogen benefits duplex alloys and many austenitic grades. Resistance of austenitic alloys depends upon nickel and sometimes nitrogen to stabilize the austenite phase and prevent precipitation of sigma or other deleterious phases.
The position of Carpenter Technology Nickel-Copper 400 alloy in this diagram can vary widely with the environment. While nickel-copper alloys offer useful resistance to many salt solutions, they are not resistant to most oxidizing salts, such as ferric chloride.
Chloride-Stress-Corrosion Cracking
A chart summarizing the resistance of various alloys to chloride-stress-corrosion cracking appears in Fig. 6. Resistance to this form of attack is strongly affected by nickel content. Resistance decreases as nickel is increased from residual to about 8%, then increases with further nickel additions to about 45%. Alloys with about 25% nickel resist cracking in many service environments. Alloys with more than about 32% nickel can be considered for more severe applications.
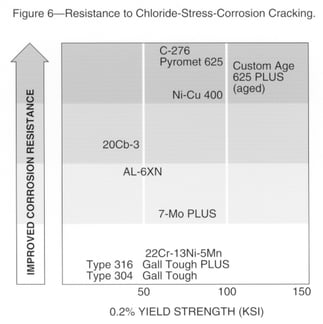
Stainless steels with increased molybdenum have provided improved resistance to chloride-stress-corrosion cracking in environments such as aqueous sodium chloride. Service experience has shown that duplex stainless grades have provided more resistance than that of Carpenter Technology 316 stainless steel. Care must be taken when welding duplex stainless steels to maintain corrosion resistance and mechanical properties.
Impure Acid Environments
Service environments often contain impurities, such as chlorides or fluorides, which can accelerate general corrosion or cause localized attack. Wet-process phosphoric acid is one example of such an environment. Fig. 7 shows relative resistance of several alloys in this environment. Resistance to general corrosion, pitting and crevice attack is important.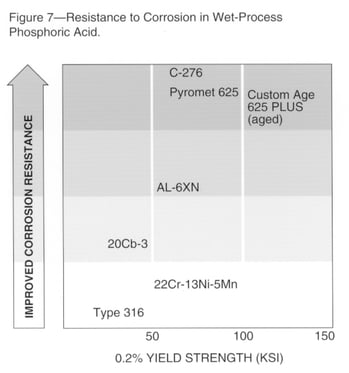
The nickel-copper alloys and Carpenter Technology 7-Mo PLUS stainless may be considered for service in various impure acid environments. The resistance of these alloys may vary depending upon the specific media. The Carpenter Technology 7-Mo PLUS stainless may be considered for oxidizing media, and the nickel-copper grades for reducing environments.
Carpenter Technology C-276 alloy (UNS N10276) offers corrosion resistance and may be considered for use in acids containing chlorides such as those found in flue gas scrubber systems. The alloy also has been used in downhole applications in oil and gas production.
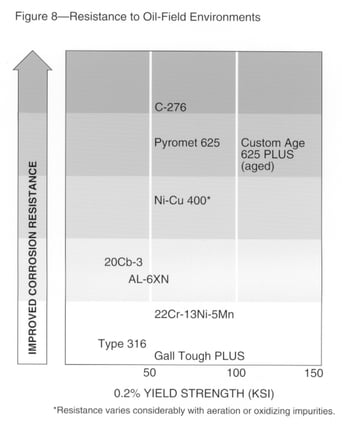
Oil Field Environments
Alloys for oil field environments must resist sulfide stress cracking, or cracking in the presence of hydrogen sulfides and water in combination with a tensile stress. High-strength, highly alloyed materials such as nickel-base alloys are susceptible to this form of attack when coupled to iron, indicating that failures are a result of hydrogen entering the metal. This type of failure can be most severe at temperatures close to ambient.
Elevated temperature cracking also can occur in the oil field in hot down-hole environments. Temperatures can be 350ºF (177ºC) or above in the presence of brine, carbon dioxide, hydrogen sulfide and possibly elemental sulfur.
Optimum resistance to both forms of cracking requires careful alloy design. A given alloy, or alloy element addition, may not provide the best resistance to both forms of cracking. Yet alloys have been used in oil fields to resist both these forms of cracking.
Resistance to pitting or crevice attack also can be required because pits may concentrate stresses. Fig. 8 shows the relative resistance of several materials to oil field environments, considering all these forms of corrosion.
Summary
Five important criteria should be weighed in selecting alloys for severely corrosive environments in the chemical process industry:
- Corrosion resistance - the most cost effective alloy will provide the corrosion resistance needed for the application without the needless expense of over-alloying. When evaluating the potential for degradation by one or more forms of corrosion, consider all details of the environment including impurities, aeration and velocity.
- Mechanical Properties - determine the need for properties such as hardness, impact resistance, fatigue strength or stress rupture resistance. Then consider the effect of the desired mechanical property(ies) on the material's corrosion resistance.
- Fabrication - Consider both design and fabrication. Contemplate the fabrication ease or difficulty, and determine the effect of fabrication on the alloy's corrosion resistance.
- Availability - Selection may be limited by availability of the candidate alloy. It may be wise to investigate before proceeding too far.
- Life Cycle Cost - Make a bottom line value analysis of the prospect alloy taking into consideration the alloy price, fabrication costs, installed cost and effective life expectancy of the finished product. Factor in maintenance and replacement costs, the associated loss of productivity, and the price of late deliveries. In many cases, the more suitable and more expensive alloy will account for only a small fraction of the total design life cycle cost.
* * *
Robert S. Brown received his B. S. in metallurgical engineering from Lafayette College, Easton, PA, in 1966. His areas of expertise lie in the corrosion resistance of stainless steels, and in welding. He has covered both subjects extensively in articles written for publication in engineering and professional journals. The author is a member and certified corrosion specialist of the National Association of Corrosion Engineers, and a member of the American Welding Society. At time of publication, he was a product application manager specializing in the chemical process, medical and energy industries at Carpenter Technology Corp., Reading, PA.